A new pathway for hydroamination. Mechanism of palladium-catalyzed addition of anilines to vinylarenes
J. Am. Chem. Soc., 2002, 124 (1166-1167)
View on publisher site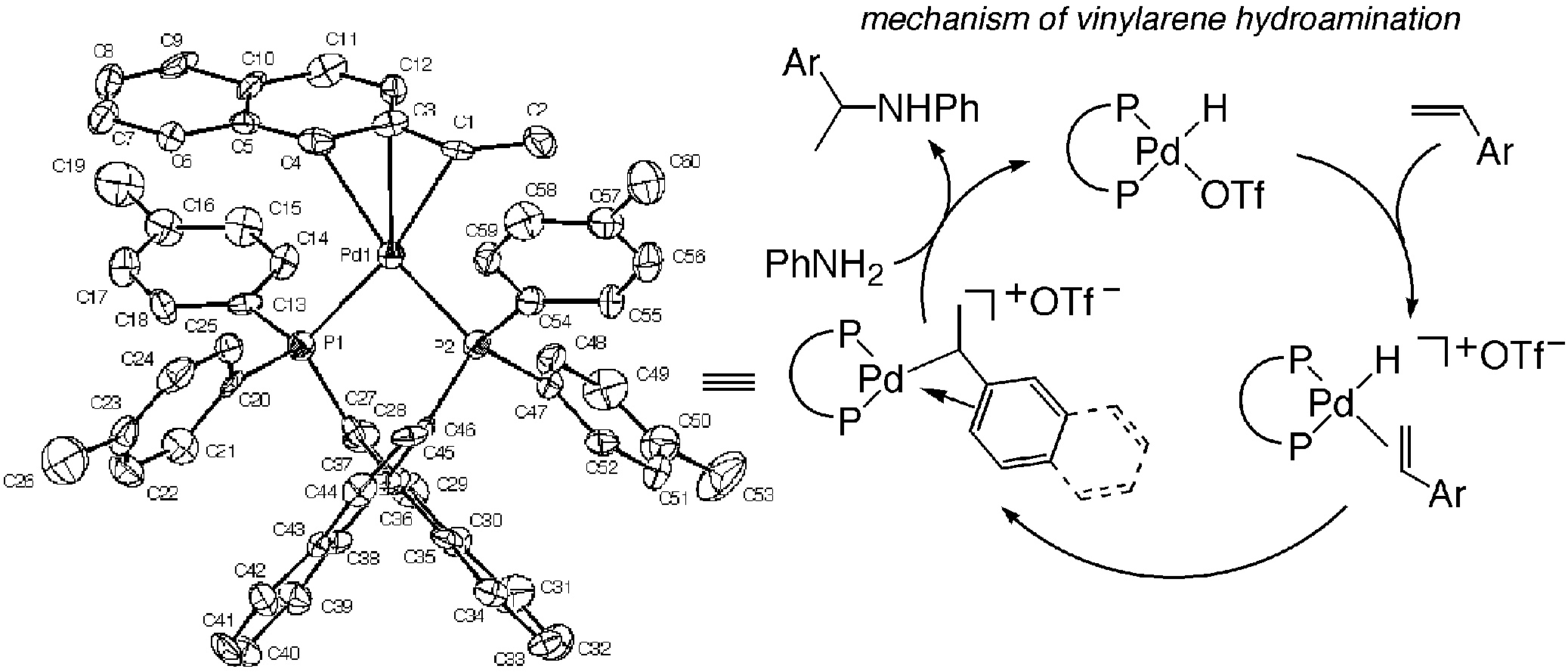
Abstract
The mechanism of the hydroamination of vinylarenes with anilines catalyzed by phosphine-ligated palladium triflates was uncovered. η3-Arylethyl diphosphine palladium triflate complexes, which result from migratory insertion of vinylarene into a palladium hydride triflate, were shown to be the resting state of the catalytic cycle. A series of these complexes has been isolated and fully characterized. The [(R)-Tol-BINAP][1-(2-naphthyl)ethyl]palladium triflate derivative 1a was crystallographically characterized. This complex reacted with aniline to form the N-phenethylaniline product in 83% yield. Reaction of the benzylic derivative [(R)-Tol-BINAP](η3-benzyl)palladium triflate with aniline also formed the N-benzylaniline product in a high 87% yield. Predominant inversion of configuration from the reaction between 1a, which is enantiopure, and aniline showed that external attack of the amine on the η3-arylethyl ligand occurred to form the product. This product from reaction of aniline with 1a is the opposite enantiomer to that obtained from the catalytic process. Thus, a minor diastereomer gives the major enantiomer in the catalytic cycle, and the major diastereomer gives the minor enantiomer. Consistent with this assertion, kinetic studies showed that the major diastereomer formed product with a rate constant roughly 3.5 times slower than the rate constant for the catalytic process that contains all diastereomers.
Read on publisher's site