Bronsted acid-catalyzed intramolecular hydroamination of protected alkenylamines. Synthesis of pyrrolidines and piperidines
Org. Lett., 2002, 4 (1471-1474)
View on publisher site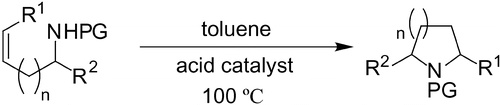
Abstract
The cyclization of aminoalkenes bearing an electron-withdrawing group on the nitrogen atom was catalyzed by triflic or sulfuric acid in toluene. Pyrrolidines and piperidines were formed in excellent yields. N-Phenylanilides also underwent cyclization to form γ-lactams.
Read on publisher's site