Site Selective Chlorination of C(sp3)-H Bonds Suitable for Late-Stage Functionalization
Angew. Chem. Int. Ed., 2021, 60 (8276-8283)
View on publisher site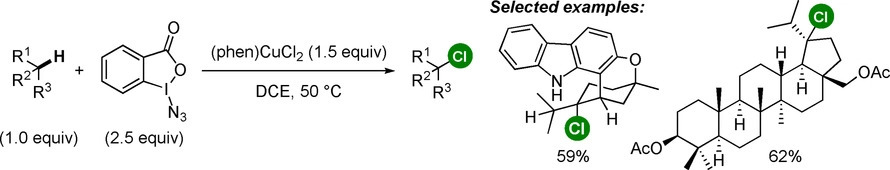
Abstract
C(sp3)−Cl bonds are present in numerous biologically active small molecules, and an ideal route for their preparation is by the chlorination of a C(sp3)−H bond. However, most current methods for the chlorination of C(sp3)−H bonds are insufficiently site selective and tolerant of functional groups to be applicable to the late-stage functionalization of complex molecules. We report a method for the highly selective chlorination of tertiary and benzylic C(sp)−H bonds to produce the corresponding chlorides, generally in high yields. The reaction occurs with a mixture of an azidoiodinane, which generates a selective H-atom abstractor under mild conditions, and a readily-accessible and inexpensive copper(II) chloride complex, which efficiently transfers a chlorine atom. The reaction's exceptional functional group tolerance is demonstrated by the chlorination of >30 diversely functionalized substrates and the late-stage chlorination of a dozen derivatives of natural products and active pharmaceutical ingredients.
Read on publisher's site