Oxalohydrazide Ligands for Copper-Catalyzed C−O Coupling Reactions with High Turnover Numbers
Angew. Chem. Int. Ed., 2021, 60 (8203-8211)
View on publisher site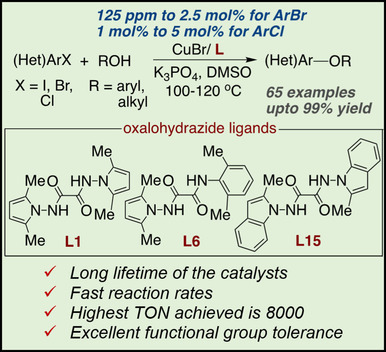
Abstract
Here, we report a class of ligands based on oxalohydrazide cores and N-amino pyrrole and N-amino indole units that generates long-lived copper catalysts for couplings that form the C−O bonds in biaryl ethers. These Cu-catalyzed coupling of phenols with aryl bromides occurred with turnovers up to 8000, a value which is nearly two orders of magnitude higher than those of prior couplings to form biaryl ethers and nearly an order of magnitude higher than those of any prior copper-catalyzed coupling of aryl bromides and chlorides. This ligand also led to copper systems that catalyze the coupling of aryl chlorides with phenols and the coupling of aryl bromides and iodides with primary benzylic and aliphatic alcohols. A wide variety of functional groups including nitriles, halides, ethers, ketones, amines, esters, amides, vinylarenes, alcohols and boronic acid esters were tolerated, and reactions occurred with aryl bromides in pharmaceutically related structures.
Read on publisher's site