Effect of Ligand Structure on the Electron Density and Activity of Iridium Catalysts for the Borylation of Alkanes
ACS Catal., 2020, 10 (3415-3424)
View on publisher site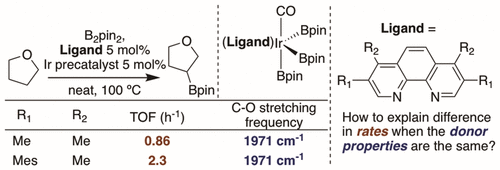
Abstract
An in-depth study of iridium catalysts for the borylation of alkyl C–H bonds is reported. Although the borylation of aryl C–H bonds can be catalyzed by iridium complexes containing phen or bpy ligands at mild temperatures and with limiting arene, the borylation of alkyl C–H bonds remains underdeveloped. We prepared a library of phenanthrolines that contain varying substitution patterns. The corresponding phen–Ir trisboryl carbon monoxide complexes were synthesized to determine the electron-donating ability of these ligands, and the initial rates for the borylation of the C–H bonds in THF and diethoxyethane β to oxygen catalyzed by Ir complexes containing these ligands were measured. For some subsets of these ligands, the donor ability correlated positively with the rate of C–H borylation catalyzed by the complexes containing ligands within a given subset. However, across subsets, ligands possessing similar donor properties to one another form catalysts for the borylation of alkyl C–H bonds with widely varying activity. This phenomenon was investigated computationally, and it was discovered that the stabilizing interactions between the phenanthroline ligand and the boryl ligands attached to Ir in the transition state for C–H oxidative addition could account for the differences in the activity of the catalysts that possess similar electron densities at Ir. The effect of these interactions on the borylation of secondary alkyl C–H bonds is larger than it is on the borylation of primary alkyl C–H bonds.
Read on publisher's site