Enantioselective α-functionaliations of ketones via allylic substitution of silyl enol ethers
Nat. Chem., 2019, 11 (177)
View on publisher site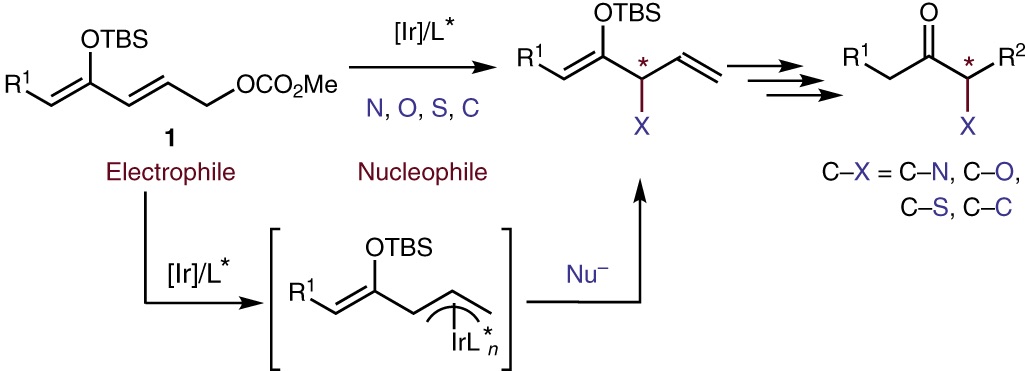
Abstract
The enantioselective construction of carbon–heteroatom and carbon–carbon bonds that are alpha to ketones leads to the formation of substructures that are ubiquitous in natural products, pharmaceuticals and agrochemicals. Traditional methods to form such bonds have relied on combining ketone enolates with electrophiles. Reactions with heteroatom-based electrophiles require special reagents in which the heteroatom, which is typically nucleophilic, has been rendered electrophilic by changes to the oxidation state. The resulting products usually require post-synthetic transformations to unveil the functional group in the final desired products. Moreover, different catalytic systems are typically required for the reaction of different electrophiles. Here, we report a strategy for the formal enantioselective α-functionalization of ketones to form products containing a diverse array of substituents at the alpha position with a single catalyst. This strategy involves an unusual reversal of the role of the nucleophile and electrophile to form C–N, C–O, C–S and C–C bonds from a series of masked ketone electrophiles and a wide range of conventional heteroatom and carbon nucleophiles catalysed by a metallacyclic iridium catalyst.
Read on publisher's site