Mechanistic Investigations of the Hydrogenolysis of Diaryl Ethers Catalyzed by Nickel Complexes of N-Heterocyclic Carbene Ligands
J. Am. Chem. Soc., 2017, 139 (17667-17676)
View on publisher site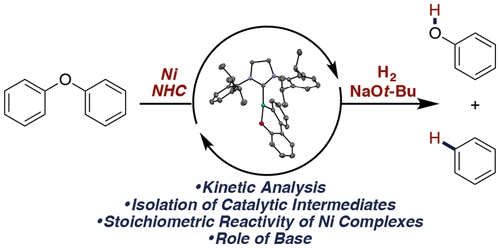
Abstract
Recent interest in the valorization of lignin has led to reactions involving the cleavage of strong aromatic C−O bonds. However, few experimental mechanistic studies of these reactions have been published. We report detailed mechanistic analysis of the hydrogenolysis of diaryl ethers catalyzed by the combination of Ni(COD)2 (COD = 1,5-cyclooctadiene) and an N-heterocyclic carbene (NHC). Experiments on the catalytic reaction indicated that NaOt-Bu was necessary for catalysis, but kinetic analysis showed that the base is not involved in the rate-limiting C−O bond cleavage. The resting state of the catalyst is an NHC-Ni(η6-arene) complex. Substitution of the coordinated solvent with diaryl ether allowed isolation of a diaryl ether-bound Ni complex. Rate-limiting C−O bond cleavage occurs to generate a three-coordinate product of oxidative addition, a metallacyclic version of which has been prepared independently. Stoichiometric studies show that arene and phenol products are released following reaction with H2. NaOt-Bu was found to deprotonate the phenol product and to prevent formation of inactive NiI dimers.
Read on publisher's site