Nickel-catalyzed Asymmetric α-Arylation and Heteroarylation of Ketones with Chloroarenes: Effect of Halide on Selectivity, Oxidation State and Room Temperature Reactions
J. Am. Chem. Soc, 2011, 133 (16330-16333)
View on publisher site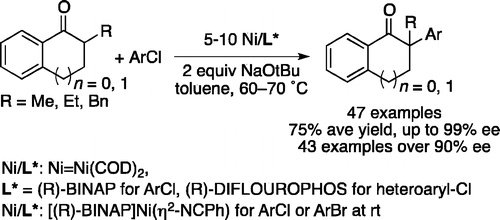
Abstract
We report the α-arylation of ketones with a range of aryl chlorides with enantioselectivities from 90 to 99% ee catalyzed by the combination of Ni(COD)2 and (R)-BINAP and the coupling of ketones with a range of heteroaryl chlorides with enantioselectivities up to 99% ee catalyzed by Ni(COD)2 and (R)-DIFLUORPHOS. The analogous reactions of bromoarenes occur with much lower enantioselectivities. Mechanistic studies showed that the difference in the rates of decomposition of the arylnickel(II) halide intermediates to {[(R)-BINAP]NiX}2 likely accounts for the difference in the enantioselectivities of the reactions of bromoarenes and chloroarenes. This catalyst decomposition can be overcome by conducting the reactions with [(R)-BINAP]Ni(η2-NC-Ph) (4), which undergoes oxidative addition to haloarenes at room temperature.
Read on publisher's site