Assessment of the Intermediacy of Arylpalladium Carboxylate Complexes in the Direct Arylation of Benzene: Evidence for C-H Bond Cleavage by ‘Ligandless’ Species
J. Am. Chem. Soc, 2011, 133 (3308-3311)
View on publisher site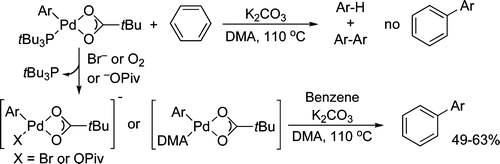
Abstract
Palladium-catalyzed direct arylations of benzene have been proposed to occur by the generation of a phosphine-ligated arylpalladium pivalate complex LPd(Ar)(OPiv) and reaction of this complex with benzene. We have isolated an example of the proposed intermediate and evaluated whether this complex does react with benzene to form the biaryl products of direct arylation. In contrast to the proposed mechanism, no biaryl product was formed from cleavage of the benzene C−H bond by LPd(Ar)(OPiv). However, reactions of LPd(Ar)(OPiv) with benzene and additives that displace or consume the phosphine ligand formed the arylated products in good yield, suggesting that a “ligandless” arylpalladium(II) carboxylate complex undergoes the C−H cleavage step. Consistent with this conclusion, we found that reactions catalyzed by Pd(OAc)2 without a ligand occur faster than, and with comparable selectivities to, reactions catalyzed by Pd(OAc)2 and a phosphine ligand.
Read on publisher's site