Palladium-Catalyzed Coupling of Ammonia with Aryl Chlorides, Bromides, Iodides, and Sulfonates: A General Method for the Preparation of Primary Arylamines
J. Am. Chem. Soc., 2009, 131 (11049-11061)
View on publisher site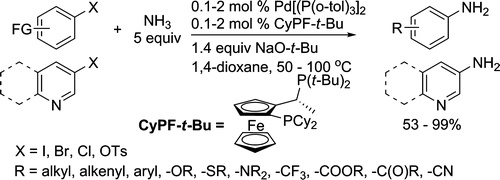
Abstract
We report that the complex generated from Pd[P(o-tol)3]2 and the alkylbisphosphine CyPF-t-Bu is a highly active and selective catalyst for the coupling of ammonia with aryl chlorides, bromides, iodides, and sulfonates. The couplings of ammonia with this catalyst conducted with a solution of ammonia in dioxane form primary arylamines from a variety of aryl electrophiles in high yields. Catalyst loadings as low as 0.1 mol % were sufficient for reactions of many aryl chlorides and bromides. In the presence of this catalyst, aryl sulfonates also coupled with ammonia for the first time in high yields. A comparison of reactions in the presence of this catalyst versus those in the presence of existing copper and palladium systems revealed a complementary, if not broader, substrate scope. The utility of this method to generate amides, imides, and carbamates is illustrated by a one-pot synthesis of a small library of these carbonyl compounds from aryl bromides and chlorides, ammonia, and acid chlorides or anhydrides. Mechanistic studies show that reactions conducted with the combination of Pd[P(o-tol)3]2 and CyPF-t-Bu as catalyst occur with faster rates and higher yields than those conducted with CyPF-t-Bu and palladiun(II) as catalyst precursors because of the low concentration of active catalyst that is generated from the combination of palladium(II), ammonia, and base.
Read on publisher's site