Effect of Ligand Steric Properties and Halide Identity on the Mechanism for Oxidative Addition of Haloarenes to Trialkylphosphine Pd(0) Complexes
J. Am. Chem. Soc., 2009, 131 (8141-8154)
View on publisher site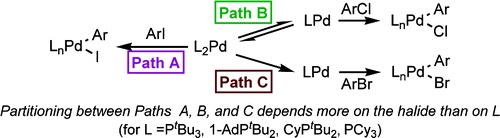
Abstract
The oxidative addition of PhX (X = I, Br, Cl) to the complexes Pd(PtBu3)2 (1), Pd(1-AdPtBu2)2 (2), Pd(CyPtBu2)2 (3), and Pd(PCy3)2 (4) (1-Ad = 1-adamantyl, Cy = cyclohexyl) was studied to determine the effect of steric properties on the coordination number of the species that undergoes oxidative addition and to determine whether the type of halide affects the identity of this species. The kinetic data imply that the number of phosphines coordinated to the complex that reacts in the irreversible step of the oxidative addition process for complexes 1−4 depends more on the halide than on the steric properties of the ligands. The rate-limiting step of the oxidative addition of PhI occurred with L2Pd(0) in all cases, as determined by the lack of dependence of kobs on [PtBu3], [1-AdPtBu2], or [CyPtBu2] and the inverse dependence of the rate constant on [PCy3] when the reaction was initiated with Pd(PCy3)3. The irreversible step of the oxidative addition of PhCl occurred with a monophosphine species in each case, as signaled by an inverse dependence of the rate constant on the concentration of ligand. The irreversible step of the oxidative addition of PhBr occurred with a bisphosphine species, as signaled by the zeroth-order or small dependence of the rate constant on the concentration of phosphine. Thus, the additions of the less reactive chloroarenes occur through lower-coordinate intermediates than additions of the more reactive haloarenes.
Read on publisher's site