Autocatalytic Oxidative Addition of PhBr to Pd(PtBu3)2 via Pd(PtBu3)2(H)(Br).
J. Am. Chem. Soc., 2008, 130 (5842-5843)
View on publisher site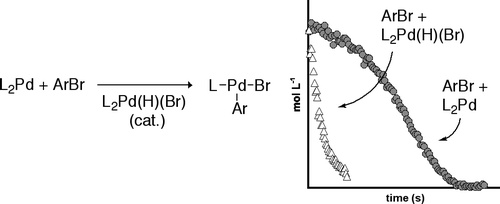
Abstract
We report that oxidative addition of bromobenzene to Pd(PtBu3)2 occurs by an unusual autocatalytic mechanism. Studies on the effect of various additives showed that the degree of rate acceleration followed the trend: (PtBu3)Pd(Ph)(Br) ≈ (HPtBu3)Br < [(PtBu3)Pd(μ-Br)]2 < (PtBu3)2Pd(H)(Br). Studies on the reactions of Pd(PtBu3)2 in the presence of (PtBu3)2Pd(H)(Br) showed that the concentration of (PtBu3)2Pd(H)(Br) decreased only after the Pd(0) complex had been consumed. These data indicated that the catalyst in this process is (PtBu3)2Pd(H)(Br). Thermal decomposition of the three-coordinate oxidative addition product (PtBu3)Pd(Ar)(Br) during the reaction of Pd(PtBu3)2 and bromoarenes ultimately leads to formation of (PtBu3)2Pd(H)(Br). Parallel reactions of bromobenzene with (PtBu3)2Pd(H)(Br) and Pd(PtBu3)2 showed that the bromoarenes reacted considerably faster with the Pd(II) species than with the Pd(0) species. We therefore propose a catalytic cycle for oxidative addition in which PBut3·HBr reacts with the Pd(0) species to form (PtBu3)2Pd(H)(Br), and (PtBu3)2Pd(H)(Br) reacts with the bromoarene, possibly though the anionic species [HPtBu3+][(PtBu3)Pd(Br)−], to form [Pd(PtBu3)(Ar)(Br)].
Read on publisher's site