Regioselective and Enantioselective Iridium-Catalyzed Allylation of Enamines
J. Am. Chem. Soc., 2007, 129 (7720-7721)
View on publisher site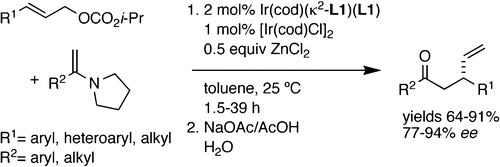
Abstract
A highly enantioselective and regioselective monoallylation of the enamines of methyl ketones has been developed. After hydrolysis of the enamine (NaOAc/AcOH(aq)), the desired allylated ketones were obtained in high yields (64−91% isolated yields), with high branched-to-linear selectivity (all but two ≥95:5), and with high enantioselectivities (all but two ≥94% ee). The reactions occur within hours at room temperature and encompass the reactions of aromatic and aliphatic methyl ketone derivatives. The enamines of methyl alkyl ketones, which exist as a mixture of two regioisomers, reacted selectively (≥99:1) at the less hindered position. The use of a preformed cyclometalated iridium catalyst, isopropyl carbonates, toluene solvent, and ZnCl2 as an alcohol adsorbent was important for the development of this process.
Read on publisher's site