Reductive Elimination of Aryl Halides upon Addition of Hindered Alkylphosphines to Dimeric Arylpalladium(II) Halide Complexes
Organometallics, 2004, 23 (1533-1541)
View on publisher site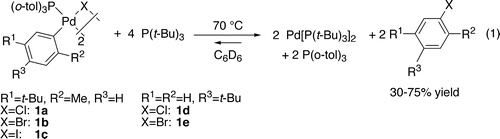
Abstract
We report the reductive elimination of haloarene from {Pd[P(o-tol)3](Ar)(μ-X)}2 (X = Cl, Br, I) upon addition of the strongly electron-donating, but sterically hindered, phosphine P(t-Bu)3 and related ligands. Reductive elimination of aryl chlorides, bromides, and iodides from these dimeric arylpalladium(II) halide complexes was observed upon the addition of P(t-Bu)3. Conditions to observe the elimination and addition equilibria were established for all three halides, and values for these equilibrium constants were measured. Reductive elimination of aryl chlorides was most favored thermodynamically, and elimination of aryl iodide was the least favored. However, reactions of the aryl chloride complexes were the slowest. Detailed mechanistic data revealed that cleavage of the starting dimer, accompanied by ligand substitution either before or after cleavage, led to the formation of a three-coordinate arylpalladium(II) halide monomer that reductively eliminated haloarene.
Read on publisher's site