Rhodium-Catalyzed Anti-Markovnikov Hydroamination of Vinylarenes
J. Am. Chem. Soc., 2003, 125 (5608-5609)
View on publisher site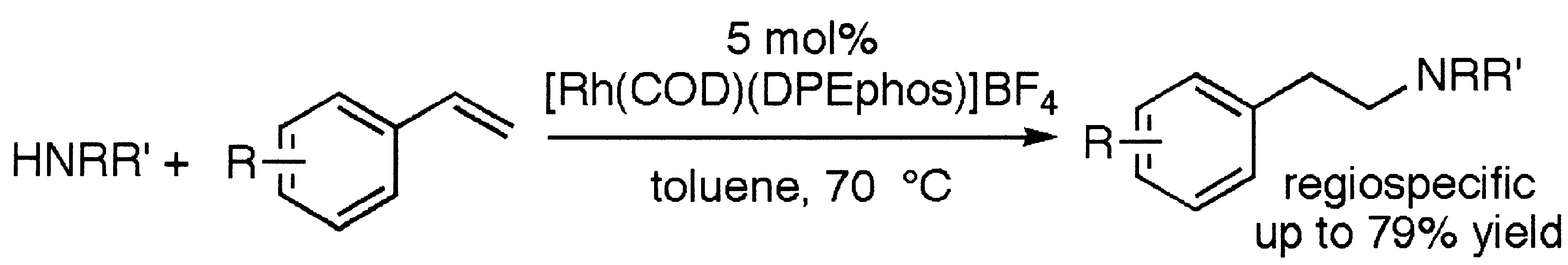
Abstract
The transition metal-catalyzed anti-Markovnikov hydroamination of unactivated vinylarenes with a rhodium complex of DPEphos is reported. The reaction of electron−neutral or electron-rich vinylarenes with a variety of secondary amines in the presence of catalyst forms the products from anti-Markovnikov hydroamination in high yields. Reactions of morpholine, N-phenylpiperazine, N-Boc-piperazine, piperidine, 2,5-dimethylmorpholine, and perhydroisoquinoline reacted with styrene to form the amine product in 51−71% yield. Reactions of a variety of vinylarenes with morpholine generated amine as the major product. Reactions of morpholine with electron-poor vinylarenes gave lower amine:enamine ratios than reactions of electron-rich vinylarenes at the same concentration of vinylarene, but conditions were developed with lower concentrations of electron-poor vinylarene to maintain formation of the amine as the major product. Reactions of dimethylamine with vinylarenes were fast and formed amine as the major product. Mechanistic studies on the hydroamination process showed that the amine:enamine ratio was lower for reactions conducted with higher concentrations of vinylarene and that one vinylarene influences the selectivity for reaction of another. A mechanism proceeding through a metallacyclic intermediate that opens in the presence of a second vinylarene accounts for these and other mechanistic observations.
Read on publisher's site