Intermolecular, Markovnikov Hydroamination of Vinylarenes with Alkylamines
J. Am. Chem. Soc., 2003, 125 (14286-14287)
View on publisher site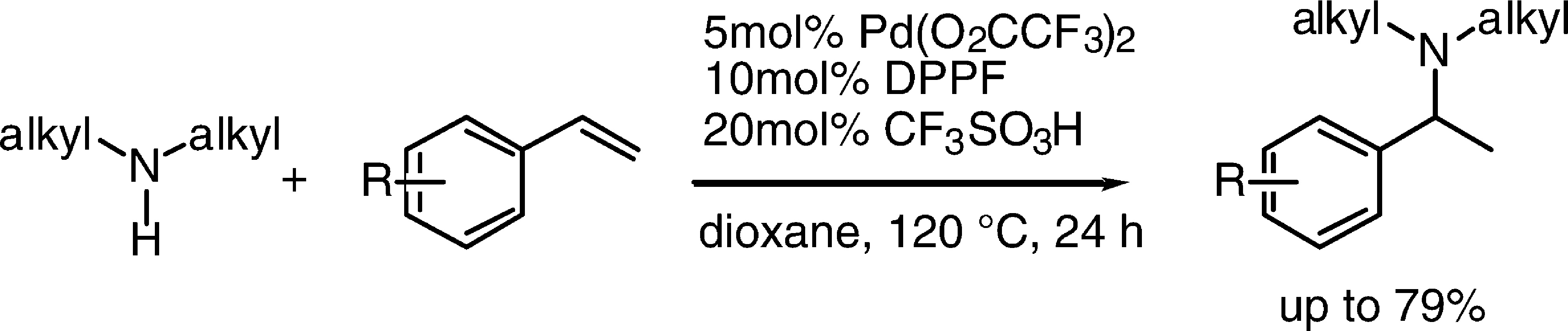
Abstract
A transition metal-catalyzed intermolecular hydroamination of vinylarenes with alkylamines is reported. The combination of Pd(O2CCF3)4, DPPF, and TfOH was the most effective catalyst of those tested. Control experiments without palladium, acid, or ligand all occurred in low yield. The reaction of various vinylarenes with cyclic and acyclic alkylamines in the presence of 5 mol % of this catalyst formed the corresponding arylethylamine products in moderate to high yields. For example, reactions of morpholine, 4-phenylpiperazine, 4-Boc-piperazine, isoindoline, and tetrahydroisoquinoline with styrene all occurred in 58−75% yield. Acyclic amines such as N-benzylmethylamine and n-hexylmethylamine reacted with 2-vinylnaphthalene in 63% and 53% yields, respectively. Mechanistic investigations showed that the reaction occurred through an η3-arylethyl palladium complex. The reactions of this complex with alkylamines generated product in combination with regenerating free vinylarene, Pd(0), and ammonium salt. Thus, one hurdle to developing hydroamination of vinylarenes with palladium complexes is the faster elimination of free vinylarene from the η3-arylethyl complex than addition to form the C−N bond. The feasibility of conducting enantioselective hydroaminations with alkylamines was also examined. The product from addition of N-benzylmethylamine to 2-vinylnaphthalene was generated in 63% ee and 36% yield in the presence of Pd(OCOCF3)2, a ferrotane ligand, and TfOH cocatalyst.
Read on publisher's site