Mechanistic Study of β-Hydrogen Elimination from Organoplatinum(II) Enolate Complexes
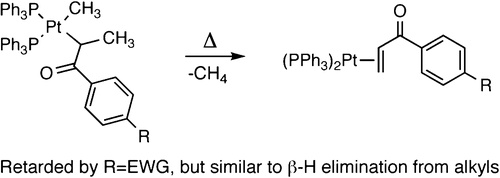
A detailed mechanistic investigation of the thermal reactions of a series of bisphosphine alkylplatinum(II) enolate complexes is reported. The reactions of methylplatinum enolate complexes in the presence of added phosphine form methane and either free or coordinated enone, depending on the steric properties of the enone. Kinetic studies were conducted to determine the relationship between the rates and mechanism of β-hydrogen elimination from enolate complexes and the rates and mechanism of β-hydrogen elimination from alkyl complexes. The rates of reactions of the enolate complexes were inversely dependent on the concentration of added phosphine, indicating that β-hydrogen elimination from the enolate complexes occurs after reversible dissociation of a phosphine. A normal, primary kinetic isotope effect was measured, and this effect was consistent with rate-limiting β-hydrogen elimination or C−H bond-forming reductive elimination to form methane. Reactions of substituted enolate complexes were also studied to determine the effect of the steric and electronic properties of the enolate complexes on the rates of β-hydrogen elimination. These studies showed that reactions of the alkylplatinum enolate complexes were retarded by electron-withdrawing substituents on the enolate and that reactions of enolate complexes possessing alkyl substituents at the β-position occurred at rates that were similar to those of complexes lacking alkyl substituents at this position. Despite the trend in electronic effects on the rates of reactions of enolate complexes and the substantial electronic differences between an enolate and an alkyl ligand, the rates of decomposition of the enolate complexes were similar to those of the analogous alkyl complexes. To the extent that the rates of reaction of the two types of complexes are different, those involving β-hydrogen elimination from the enolate ligand were faster. A difference between the rate-determining steps for decomposition of the two classes of complexes and an effect of stereochemistry on the selectivity for β-hydrogen elimination are possible origins of the observed phenomena.
Read more on publisher's site.