Palladium-Catalyzed Cross Coupling of Secondary and Tertiary Alkyl Bromides with a Nitrogen Nucleophile
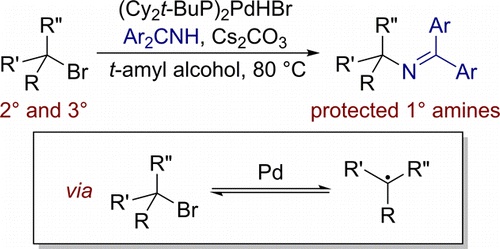
We report a new class of catalytic reaction: the thermal substitution of a secondary and or tertiary alkyl halide with a nitrogen nucleophile. The alkylation of a nitrogen nucleophile with an alkyl halide is a classical method for the construction of C–N bonds, but traditional substitution reactions are challenging to achieve with a secondary and or tertiary alkyl electrophile due to competing elimination reactions. A catalytic process could address this limitation, but thermal, catalytic coupling of alkyl halides with a nitrogen nucleophile and any type of catalytic coupling of an unactivated tertiary alkyl halide with a nitrogen nucleophile are unknown. We report the coupling of unactivated secondary and tertiary alkyl bromides with benzophenone imines to produce protected primary amines in the presence of palladium ligated by the hindered trialkylphosphine Cy2t-BuP. Mechanistic studies indicate that this amination of alkyl halides occurs by a reversible reaction to form a free alkyl radical.
Read more on publisher's site.