Enantioselective Total Syntheses of (−)-Taiwaniaquinone H and (−)-Taiwaniaquinol B by Iridium-Catalyzed Borylation and Palladium-Catalyzed Asymmetric α-Arylation
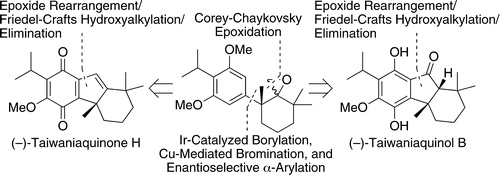
We report a concise, enantioselective total synthesis of (−)-taiwaniaquinone H and the first enantioselective total synthesis of (−)-taiwaniaquinol B by a route that includes asymmetric palladium-catalyzed α-arylation of a ketone with an aryl bromide that was generated by sterically controlled halogenation via iridium-catalyzed C−H borylation. This asymmetric α-arylation creates the benzylic quaternary stereogenic center present in the taiwaniaquinoids. The synthesis was completed efficiently by developing a Lewis acid-promoted cascade to construct the [6,5,6] tricyclic core of an intermediate common to the synthesis of a number of taiwaniaquinoids. Through the preparation of these compounds, we demonstrate the utility of constructing benzylic quaternary stereogenic centers, even those lacking a carbonyl group in the α-position, by asymmetric α-arylation.
Read more on publisher's site.