Resting State and Elementary Steps of the Coupling of Aryl Halides with Thiols Catalyzed by Alkylbisphosphine Complexes of Palladium
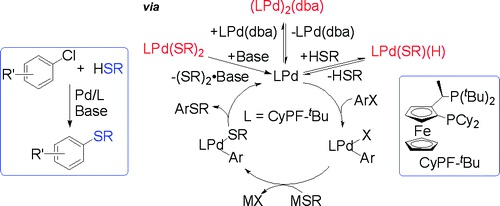
Detailed mechanistic studies on the coupling of aryl halides with thiols catalyzed by palladium complexes of the alkylbisphosphine ligand CyPF-tBu (1-dicyclohexylphosphino-2-di-tert-butylphosphinoethylferrocene) are reported. The elementary steps that constitute the catalytic cycle, i.e. oxidative addition, transmetalation and reductive elimination, have been studied, and their relative rates are reported. Each of the steps of the catalytic process occurs at temperatures that are much lower than those required for the reactions catalyzed by a combination of palladium precursors and CyPF-tBu. To explain these differences in rates between the catalytic and stoichiometric reactions, studies were conducted to identify the resting state of the catalyst of the reactions catalyzed by a combination of Pd(OAc)2 and CyPF-tBu, a combination of Pd(dba)2 and CyPF-tBu, or the likely intermediate Pd(CyPF-tBu)(Ar)(Br). These data show that the major palladium complex in each case lies off of the catalytic cycle. The resting state of the reactions catalyzed by Pd(OAc)2 and CyPF-tBu was the palladium bis-thiolate complex [Pd(CyPF-tBu)(SR)2] (R = alkyl or aryl). The resting state in reactions catalyzed by Pd2(dba)3 and CyPF-tBu was the binuclear complex [Pd(CyPF-tBu)]2(μ2,η2-dba) (9). The resting states of reactions of both aromatic and aliphatic thiols catalyzed by [Pd(CyPF-tBu)(p-tolyl)(Br)] (3a) were the hydridopalladium thiolate complexes [Pd(CyPF-tBu)(H)(SR)] (R= alkyl and aryl). All these palladium species have been prepared independently, and the mechanisms by which they enter the catalytic cycle have been examined in detail. These features of the reaction catalyzed by palladium and CyPF-tBu have been compared with those of reactions catalyzed by the alkylbisphosphine DiPPF and Pd(OAc)2 or Pd(dba)2. Our data indicate that the resting states of these reactions are similar to each other and that our mechanistic conclusions about reactions catalyzed by palladium and CyPF-tBu can be extrapolated to reactions catalyzed by complexes of other electron-rich bisphosphines.
Read more on publisher's site.